Invimeds ensure the non-discretion policy for trials to follow privacy.
Clinical Supply Management - Enhancing Seamless Clinical Trial Material Sourcing
Biological products that are highly similar to biologic drugs approved by regulatory agencies are called biosimilars – they are the generic versions of biologic drugs. Twenty-eight biologics with annual U.S. sales of more than $1 billion per year are anticipated to lose their exclusivity in the U.S. till 2029. Therefore, we foresee significant future growth opportunities for generics. Planning and managing clinical trial material supplies (CTMS) can be a challenging task. There are complex testing requirements, including head-to-head analytical/non-clinical/clinical testing against the reference originator, to ensure biosimilar quality.
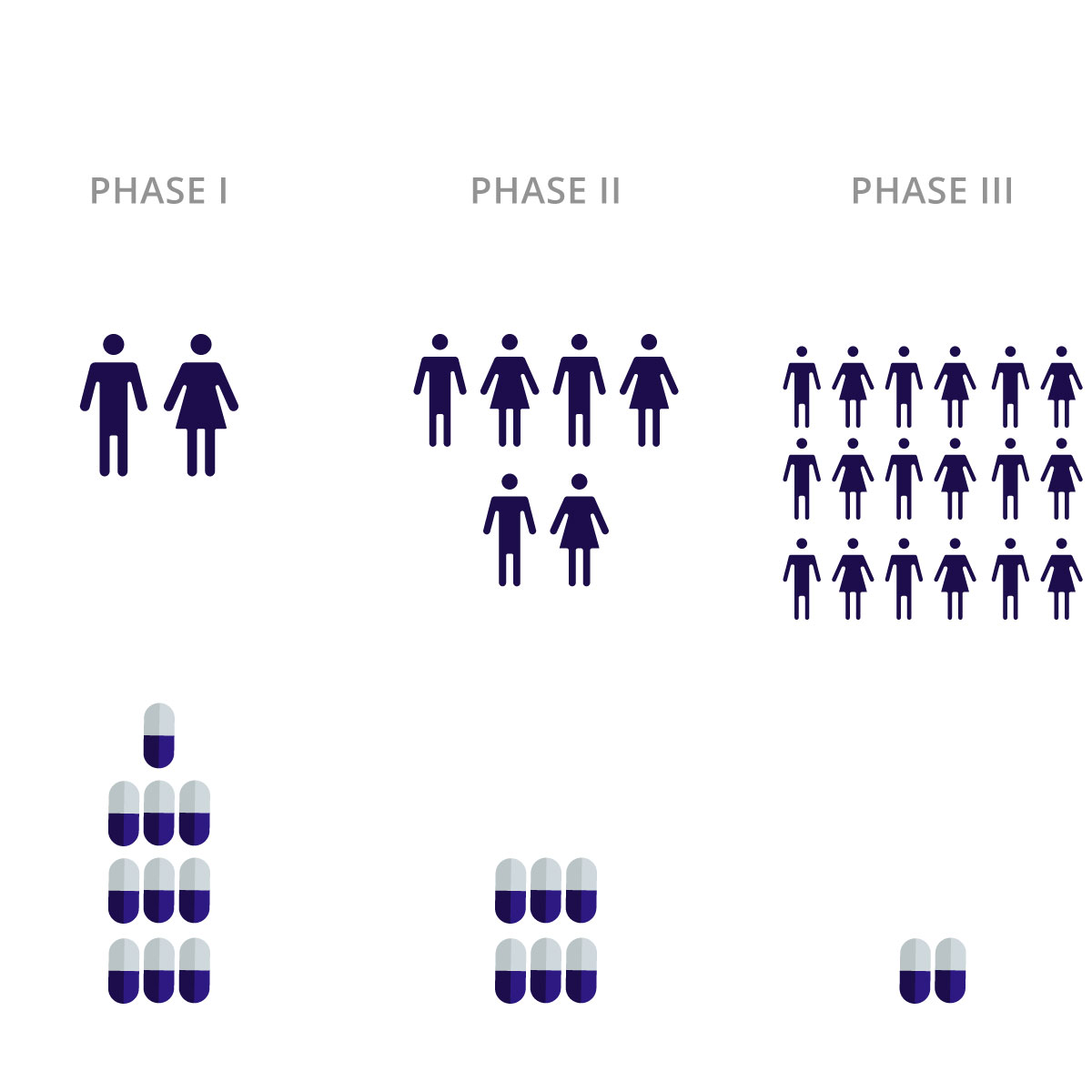
Our Services for Clinical Trial
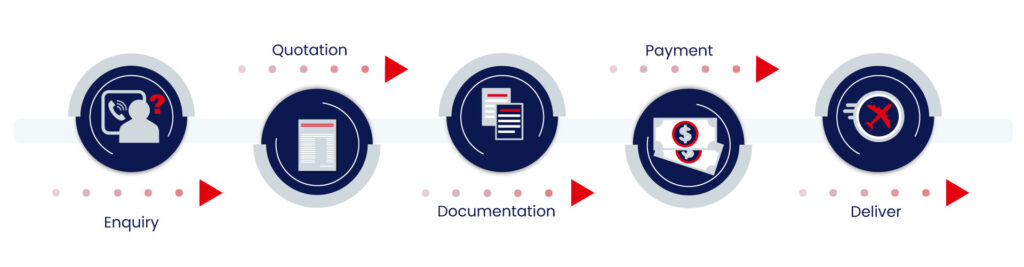
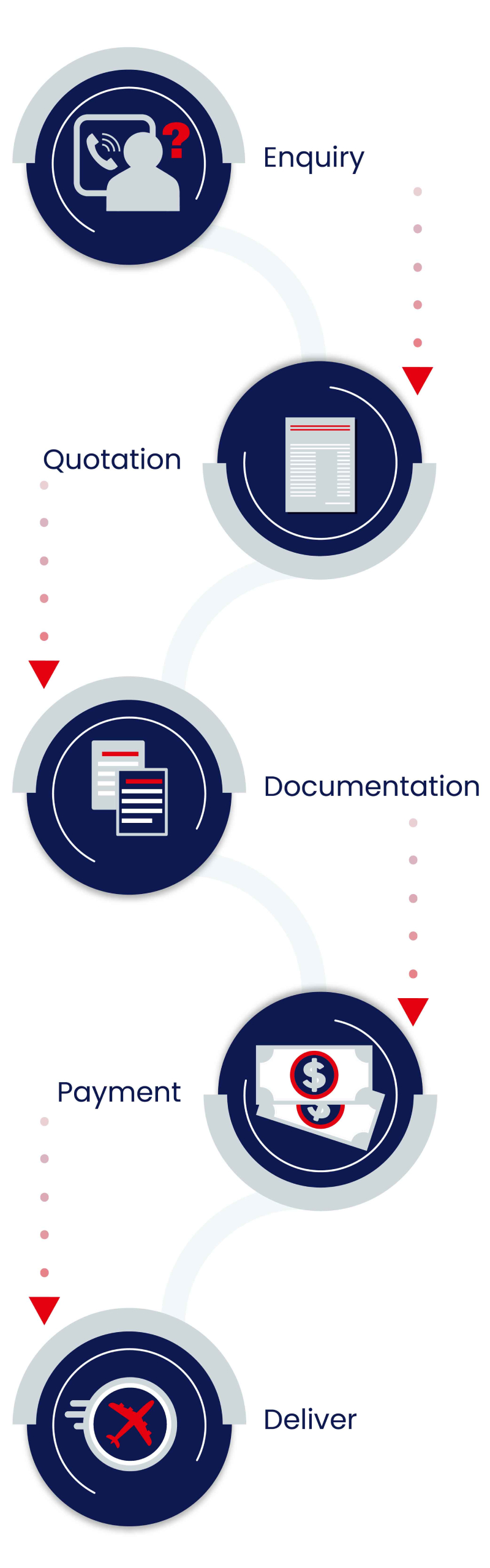
Our Process
Why you should trust invimeds for RLD?
High-Caliber Distribution
Our depot and distribution services are high caliber, dependable, and reasonably priced.
Comprehensive Warehousing Support
We provide comprehensive warehousing and logistics services to support clinical trials, maintaining the best operating standards in the industry.
Extensive Network of Depot Partners
We have a vast network of third-party depot partners and numerous depot locations for operations.
Experts in International Customs
We have expertise in handling international customs following standard regulatory procedures.
Cold-chain Shipping
Cold-chain shipping guarantees that temperature-sensitive research medications reach your clinical trial facilities within the appropriate temperature range.

Interact with experts
Our qualified Clinical Trial Supply experts are only a phone call away to guide you through the procedure of trial supplies and help you get started. We are among the leading Comparator Sourcing companies handling seamless comparator sourcing.
Invimeds Health delivers innovative and hassle-free Clinical Supply Management solutions across the globe. Speak with our experts today!
Get in touch
Have an inquiry or some feedback for us? Fill out the form below to contact our team
Latest Updates
FAQs
Are you sourcing globally?
Our Reference Listed Drugs (RLDs) are medications that regulatory bodies have approved in various nations or areas, and we ensure to source them globally while distributing them worldwide. We have exclusive production and distribution rights to promote and sell RLDs. Depending on our corporate strategy, we source the drugs locally or worldwide.
Can you obtain the documents?
Yes, we can get the documentation for the RLD product. Moreover, we have access to the paperwork that regulatory bodies need to support the approval and marketing of their medicinal effect. In addition to adhering to all regulatory standards associated with the approval, marketing, and distribution of their medicinal product, we are responsible for ensuring that their product is secure and suitable for the intended purpose.
How do you support clients through the buying cycle?
We usually help customers through the purchase cycle by giving them information and support about their product and guiding them through the logistical and regulatory hurdles involved in obtaining and utilizing a pharmaceutical product. While a consumer is making a purchase, we may offer customer care and support, including answering inquiries, giving product samples, and addressing any problems that might come up.